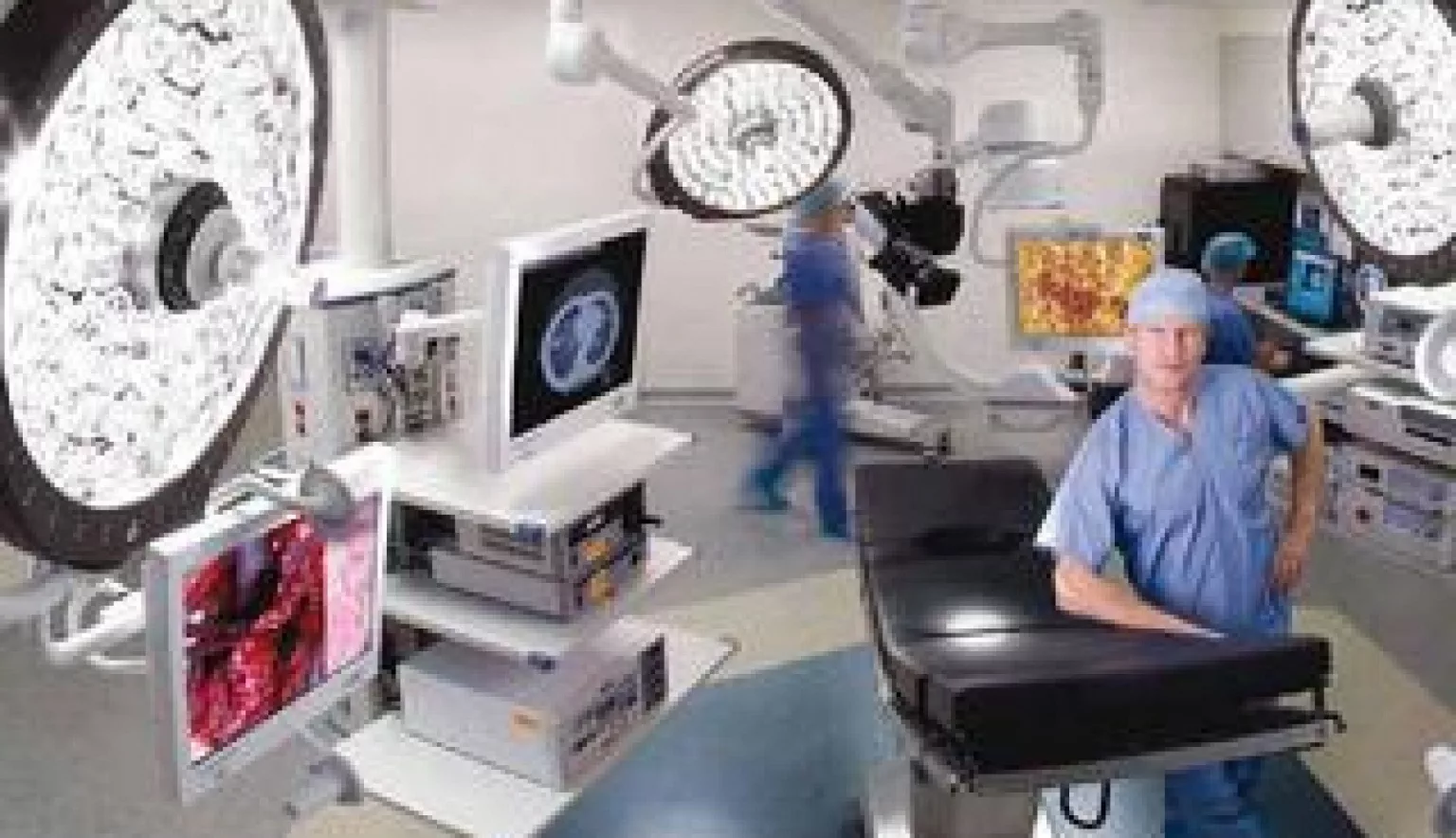
SafMed offers a range of products and services that help customers do their jobs more safely, effectively and efficiently.
SURGICAL STEEL
Founded in 1988, SafMed is a private company registered in South Africa, which specialises in the importation, production and marketing of decontamination, infection prevention and surgical products.
It has developed a strong customer base in both the private and public provincial hospital segments and has built a reputation within the industry as being market leaders within a number of product offerings.
“Our model is based around value based products with an emphasis on quality,” says Marketing Director Pat Ayling. “The sorts of products on offer include CSSD equipment and cosumables,, infection prevention products and surgical support equipment.” There are plenty of opportunities for companies like SafMed. One lies in medical instrument traceability.
“If we think back five to 10 years, looking at sterilization for example, which is probably the biggest part of our business and where we started out, we have seen that the washing of equipment has come to the fore and I think hospitals are realising that they need to control that process,” says Ayling. “Not only in this country, but worldwide, there is recognition that it is not just about the sterilizing process and that it is very important that before you sterilize instruments are properly cleaned. The other trend we have seen relates to traceability. That is coming to the fore. We are looking at introducing computerised systems for this purpose.
“The other area that we are looking at is what is happening now with surgical instrument design and how they are becoming a lot more complex and difficult to clean, sterilize, or both. It is the responsibility of the manufacturers; if they build something, they have to give clear instructions as to how those devices should be reprocessed. Some of the more complex systems need longer sterilizing times.”
Another trend is a drive for “minimally invasive” surgery (MIS). There are several benefits: with smaller incisions, there’s less trauma; most incisions just take a stitch or two to close; faster recovery; and having to spend less time in hospital – minimally invasive surgeries are associated with a 23 hour discharge or scheduled outpatient surgery.
“We’ve seen that,” says Ayling. “There is a drive for MIS and for us the growth would be in surgical table design. We’re seeing a lot of innovation here, and it is revolutionary. The old tables were stainless steel. Now they are carbon fibre. It doesn’t interfere with x-rays or imaging capabilities of the various machines looking inside the patient. The other thing, and we have been involved with one of the imaging companies on this, is hybrid operating theatres, mainly for cardiovascular work. The end result is better patient care.”
New technology means there’s also a need for “increased integration”.
“As technologies develop, you end up with lots of equipment all over the operating room floor,” says Ayling. “The latest thinking is integration, taking all these signals, from all the various monitoring equipment and routing them through a special device. The surgeon no longer needs ten different video screens to monitor the patient’s progress during the operation.”
Most of these trends are driven by the private sector; remember South Africa’s healthcare system is one divided. There’s a big public–private split.
The South African government’s proposed national insurance scheme aims to tackle this however.
“The South African Government hopes its proposed National Health Insurance (NHI) scheme will bridge the gap between public and private healthcare by providing universal access based on need rather than ability to pay,” says Ayling. “It’s being rolled out. It’ll be some years before we know if it is a success, if it works and how it can or will change the landscape. It can only be good for South Africans.
“What I understand will happen is that on the public side the government will set up an almost standard list of equipment that should be in a hospital. So the cardiac department should have certain types of equipment, particular beds etc. That is a start. They are also looking at hospital design, trying to come up with standard designs. It is an exciting time. How long it will take I don’t know. But that is what we are seeing.”
South Africa does not have a comprehensive system of medical device regulation.
However, the market is very sophisticated and it is strongly advisable that products are either CE or FDA approved.
The South African Department of Health is currently in the process of drafting the necessary policy documents, which cover the regulation of all devices. At the moment only electro-medical devices are regulated
“In South Africa regulation is a big issue so far as medical devices and equipment goes. At present it is not regulated at all aside from electro medical devices so virtually anything can be sold. It doesn’t necessarily mean it will be brought but it certainly can come into the market. There has been a push to try and get regulation for many years. That is on the cards as well. It will have implications going forward. Again, it is difficult to speculate as to when things will happen. But I think it will.”
Ayling says there is potential to expand into neighbouring countries and further across Africa.
“We do some work in the close territories – Namibia, Botswana, Lesotho and Mozambique,” he says. “But we don’t do it direct. It is via distributors and certainly we are looking at it. Unfortunately the biggest challenge is the level to which the market is developed. They will develop as growth takes place – and these are fast growing economies – but, at the moment, it is still very basic healthcare in a lot of countries. That does present opportunity. But education is also important. There needs to be an acceptance of the importance of the right devices and equipment.
“Our challenge, apart from economic ones, is being paid. I think there are plenty of horror stories. A lot of funds coming into Central Africa are donor funds still so they might place orders and then not pay because they are waiting for donor funds. We are adopting a watch and see approach. It is an area we keep an eye on and we are seeing some of the private hospital groups expanding into this market. We worked with a private hospital in Botswana and last year we worked with Netcare on a PPP in Lesotho. We look at opportunities like that.”
SafMed has been working with high performance materials company Ahlstrom in the area of sterilization wraps. This is another growth area says Ayling. “The purpose of wrapping is to keep something sterile for whenever it needs to be used for, without contamination,” he explains. “The market trend moved initially towards paper-based solutions in South Africa and then moved onto non-woven type products, which are a combination of paper and polymers. We have been working with Ahlstrom who have come up with a product which we are excited about. To go back a step, the challenge hospitals have at the moment is that they tend to want every conceivable instrument – the private-side of the industry that is – and they might only use a small number of them in the operation. But they want the security of having them all available in case. The challenge that you face is that when you wrap the instrument sets you need to protect the specialised packaging material otherwise it gets damaged just from the weight of the set and they are still using linen on the outside. We are working with Ahlstrom on a product that is a combination of non-woven and not linen. You don’t want to use linen because it is expensive and also tends to give off a lot of fibre in an operating theatre and that’s not desirable. But to get the market to accept concepts like that – this is just an example – takes a lot of work. Eventually we get there.”
SafMed has a manufacturing facility in Cape Town where it makes its own branded products.
Ayling says the company has started GS1 bar-coding all of its products, keeping track of “where the product was made, who made it, when it was made, when it expires, and any special storage conditions that you need”. Again it is about traceability. “Most of the EU and US products are bar-coded like this,” he says. “It is in its infancy in South Africa. It’ll help drive up safety and standards. There are issues of counterfeit products, expired products being used and just wrong products being used on patients. GS1 will streamline everything and make hospitals and surgeries safer and more efficient. We’re very much ahead of the game on this locally.”
To learn more about SafMed visit www.safmed.co.za.